Mar 23, 2026
Antibody Glycosylation: FDA requirements and Characterization
Fc glycosylation controls antibody efficacy and safety. Learn what FDA expects for characterization, manufacturing and biosimilar comparability.
 \
\
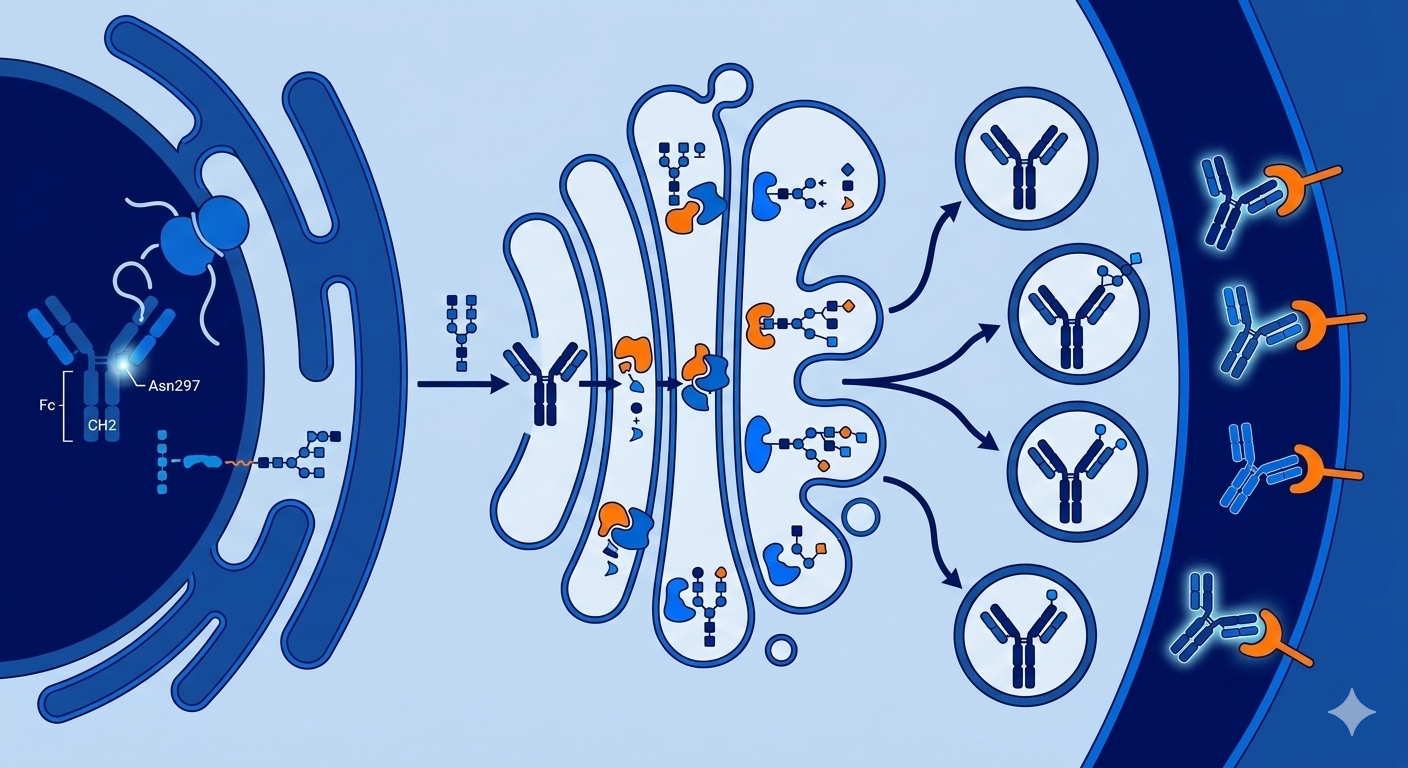
Antibody's Fc region glycosylation is one of the most consequential critical quality attributes (CQAs) in therapeutic antibody development. Regulators at both the FDA and EMA require thorough characterization, consistent batch-to-batch control, and functional bioassay correlation as part of IND and BLA submissions
What Is Antibody Glycosylation?
Glycosylation is the cellular process by which sugar molecules, known as glycans, are covalently attached to a protein backbone. In therapeutic IgG antibodies, the most critical glycosylation event is N-linked glycosylation at asparagine 297 (Asn297) in the CH2 domain of the Fc region. This single site is conserved across all four human IgG subclasses (IgG1 through IgG4).
The sugars themselves are monosaccharide units that link together in branched structures, these include: N-acetylglucosamine (GlcNAc), mannose, galactose, fucose, and sialic acid. Unlike protein synthesis, glycosylation is not template-driven. It occurs through a sequential enzymatic process in the endoplasmic reticulum and Golgi apparatus, involving glycosyltransferases and glycosidases.
Because this process lacks a genetic template, it inherently produces a mixture of glycoforms rather than a single uniform structure. This means that every batch of a therapeutic antibody contains a population of glycoforms. The relative abundance of each glycoform, and the presence or absence of specific sugar residues such as core fucose, terminal galactose, or sialic acid, determines how the antibody interacts with Fc receptors on immune cells and, by extension, how it performs clinically.
Why Glycosylation Is a Critical Quality Attribute
A critical quality attribute is defined as a physical, chemical, biological, or microbiological property that must remain within an appropriate limit, range, or distribution to ensure the desired product quality, safety, and efficacy.
For programs where the mechanism of action depends on immune effector function (including antibody-dependent cell-mediated cytotoxicity (ADCC) or complement-dependent cytotoxicity (CDC)) the glycan profile attached to the Fc region directly influences clinical performance.

Effector function
The Fc domain engages immune effector cells (natural killer (NK) cells, macrophages) as well as the complement system. The glycan structure at Asn297 fine-tunes this engagement. Core fucosylation, in particular, is the strongest single driver of ADCC activity. Antibodies lacking core fucose on the Fc N-glycan (afucosylated antibodies) bind FcγRIIIa with dramatically higher affinity than their fucosylated counterparts, enhancing ADCC by 5- to 50-fold depending on the assay system. This is why afucosylation is a closely monitored CQA in any program where ADCC is part of the mechanism of action.
Pharmacokinetics
High-mannose glycoforms are associated with accelerated serum clearance due to recognition by mannose receptors on liver cells. Terminal galactosylation and sialylation influence half-life and immune recognition. Even relatively minor changes in the distribution of galactosylated versus agalactosylated species can shift the PK behavior of a biologic. For example, the ratio of G1F to G0F.
Immunogenicity
Alpha-galactose (α-Gal), found in antibodies produced by NS0 and SP2/0 mouse myeloma cell lines, and N-glycolylneuraminic acid (Neu5Gc) are non-human glycan structures that have been associated with adverse immune reactions in patients. This is a primary reason why the industry has shifted toward Chinese hamster ovary (CHO) cell expression systems, which do not produce these non-human epitopes.
Manufacturing consistency
Because glycosylation is sensitive to cell culture conditions, the glycan profile can shift with changes in the expression system, cell bank passage, bioreactor parameters (pH, dissolved oxygen, temperature), nutrient feed composition, and even storage conditions. This sensitivity makes glycan control a manufacturing challenge, not just an analytical one.
The regulatory implication is clear: glycan analysis must be integrated into both characterization and routine batch release testing, supported by robust in-process controls and, where scientifically justified, functional bioassays.
What the FDA Has Observed Across Approved Antibodies
Recent benchmark work by FDA scientists at the Center for Drug Evaluation and Research (CDER), presented at FDA Grand Rounds in 2026, examined glycan profiles across the full landscape of FDA-approved therapeutic antibodies.
As of May 2025, the FDA has approved 209 therapeutic antibodies covering oncology, autoimmune disease, cardiovascular conditions, and infectious disease. The majority (71%) are IgG1, and 81% are produced in CHO cell expression systems.
Analysis of 123 CHO-produced antibodies revealed that the top 10 most abundant glycoforms account for over 99% of total glycans across approved products. The three dominant glycoforms are G0F, G1F, and G2F.
While the same glycoforms appear consistently across products, their relative abundances vary widely. G0F alone ranges from approximately 20% to 90% depending on the product. This variation is a direct consequence of how FDA sets specifications: acceptance criteria are defined based on each molecule's mechanism of action, its clinical context, and the manufacturing process variability demonstrated during development, rather than against a universal industry standard.
For antibodies with relevant effector function, FDA has consistently observed five afucosylated species controlled in drug substance release specifications: G0, G1, G2, G0-GlcNAc, and Man5. Acceptance ranges for total afucosylation vary from as narrow as 0.5%–1.7% to as broad as 0%–12%, depending on the product's clinical relevance and mechanism.
Regarding antibodies produced in NS0 and SP2/0 mice cell lines, the analysis confirmed the presence of non-human glycans (α-Gal in NS0, Neu5Gc in SP2/0), which is why several biosimilar manufacturers have switched to CHO expression systems specifically to eliminate these epitopes.
For biosimilars products, the FDA data confirmed that approved biosimilar products share virtually identical top-10 glycan profiles compared to their reference products, reinforcing the regulatory expectation that glycan comparability is a cornerstone of biosimilarity demonstration.
Analysis of product labeling across all 41 antibodies that describe Fc effector functions as part of their mechanism of action revealed a consistent pattern: ADCC and CDC are described exclusively using qualifiers such as "in vitro," "proposed mechanism of action," or "possible mechanisms," with no product carrying a label claim supported by direct clinical validation of effector function. This reflects regulatory acknowledgment of mechanistic hypotheses derived from laboratory findings, and CMC teams should align their characterization strategy and labeling expectations accordingly.
Glycan Analytical Methods: Current Industry Practice
The glycan analysis framework expected by regulators is documented primarily in ICH Q6B and ICH Q5E. Both FDA and EMA require characterization of carbohydrate content, oligosaccharide profiles, and glycosylation site occupancy. The EMA additionally specifies particular attention to sialylation, galactosylation, mannosylation, and fucosylation levels.
The standard analytical workflow involves enzymatic glycan release (typically using PNGase F), fluorescent labeling, and chromatographic separation. HILIC is the most commonly used separation method, with 2-AB labeling applied in approximately 78% of BLA submissions reviewed by FDA. RapiFluor-MS is seeing increased adoption where mass spectrometric detection is paired with fluorescence. Mass spectrometry provides essential orthogonal confirmation of glycan composition, linkage, and site occupancy, and is particularly important for afucosylation quantitation and low-abundance glycoform characterization.
46% of FDA-approved biologics include glycan analysis in drug substance release specifications. Among the 41 products that describe Fc effector functions as part of their mechanism of action, 76% use both glycan analysis and functional bioassays, 12% use glycan analysis alone, and 12% use bioassays alone. For programs where ADCC is claimed as a mechanism, the combined use of structural and functional assays reflects the regulatory totality-of-evidence expectation and is not optional.
FDA researchers have also developed a lectin microarray platform for rapid glycan epitope profiling of intact antibodies, without requiring glycan release or labeling. The technology is still in development for routine use, but it represents the direction regulators are moving and CMC teams should monitor its adoption trajectory.
Glycan Control in Biosimilar Development
Glycan comparability is one of the most technically demanding elements of the biosimilar analytical similarity exercise. Because glycosylation is not encoded in the DNA sequence, two antibodies with identical amino acid sequences can have materially different glycan profiles if the expression system or manufacturing process differs. Differences in G0F/G1F/G2F distribution, afucosylation level, or high-mannose content can all affect effector function or PK.
The regulatory question is whether any observed difference has a meaningful clinical impact on PK/PD behavior, safety, or efficacy. This is assessed through structural characterization, functional bioassays (ADCC, CDC, FcγRIII binding), and PK comparability data under the FDA totality-of-evidence framework. Glycan specifications should therefore be defined relative to the observed variability of the reference product across multiple lots, with pre-specified statistical acceptance ranges rather than a binary match/no-match judgment.
Any upstream change can alter the glycan profile. Comparability studies following process changes must include glycan analysis as a primary element, particularly for products with effector function.
This includes bioreactor conditions, media composition, cell bank passage, purification scheme:
-
Dissolved oxygen, pH and CO2 adjustments typically produce glycan index changes below 5%, and often below 1.5%.
-
Temperature and osmolality are the exception: temperature downshifts and hyperosmolality can drive galactosylation changes exceeding 10% and fucosylation changes of 1–10%, magnitudes that are biologically relevant given that even small reductions in core fucose enhance FcγRIIIa binding and ADCC activity. These two parameters deserve the tightest control windows in the manufacturing design space.
When reporting glycan data, glycan distribution and glycan indices should always be reported together. Indices alone can mask glycoform redistributions that matter for bioactivity. A shift between G1F and G2F, for example, may leave the galactosylation index unchanged while altering CDC potential.
Regulatory Expectations: What to Include in Your CMC Package
The following table summarizes key regulatory expectations for glycan characterization and control in IND and BLA submissions.
| Development Stage | Expectation |
| Pre-IND / early characterization | Glycan profile identification (released glycans, HILIC or LC-MS); site occupancy; confirmation of absence of immunogenic non-human glycans |
| IND submission | Glycan profile with quantitation of major glycoforms; in-process controls for glycosylation; preliminary acceptance ranges |
| BLA submission | Validated glycan release assay included in DS specification; ADCC/CDC bioassays where effector function is part of MOA; afucosylation control with defined acceptance range; batch-to-batch consistency data |
| Biosimilar submission | Full analytical similarity assessment vs. reference product across multiple lots; statistical acceptance criteria; functional bioassay correlation; totality of evidence narrative |
Several practical points follow from FDA's review experience across BLA submissions:
First, glycosylation specifications are product-specific. There is no single industry standard for acceptable G0F content or afucosylation range. Specifications must be anchored in the molecule's mechanism of action, its clinical context, and the manufacturing process variability demonstrated during development.
Second, product labeling for Fc effector function is typically qualified. Language such as "in vitro ADCC" or "proposed mechanism" is standard, reflecting the mechanistic hypothesis with limited direct clinical validation. This is an important distinction: the labeling describes mechanistic context, not a clinical claim. CMC teams should align their characterization strategy accordingly.
Third, the use of bioassays alongside glycan analysis is becoming standard practice for effector function molecules. For biosimilars, where demonstrating functional equivalence is a regulatory requirement, a well-designed ADCC or CDC bioassay with appropriate reference standards and acceptance criteria is as important as the glycan profile itself.
How Crystal Bio Solutions Supports Glycan Characterization and Control
At Crystal Bio Solutions (CBS), we provide end-to-end CMC analytical services for therapeutic antibodies, ADCs, and biosimilars, including comprehensive glycan profiling strategies designed to meet FDA regulatory expectations.
Our capabilities span N-glycan release and profiling using HILIC and LC-MS platforms, glycopeptide analysis, afucosylation quantitation, and cell-based functional bioassays including ADCC and CDC assays.
For biosimilar programs, we support the full analytical similarity exercise, from reference product characterization across multiple lots, to statistical acceptance criteria definition, to the integration of structural and functional data into the comparability narrative.
For IND-stage programs, we help design glycan characterization packages aligned with ICH Q6B and FDA expectations, so that the analytical strategy built in early development scales cleanly into the BLA submission package.
Frequently Asked Questions
What is a glycan, and why does it matter for therapeutic antibodies?
A glycan is a branched carbohydrate structure composed of linked sugar units. In therapeutic IgG antibodies, the glycan attached to Asn297 in the Fc region influences how the antibody interacts with immune receptors. Changes in glycan composition can alter ADCC activity, complement activation, serum half-life, and immunogenicity.
Which glycoforms are most common in FDA-approved antibodies?
Across CHO-produced antibodies, the three most abundant glycoforms are G0F, G1F, and G2F (all core-fucosylated, complex-type biantennary structures). Together they account for approximately 88% of total glycans. However, the exact distribution varies significantly between products.
What is afucosylation, and why is it important?
Afucosylation refers to the absence of core fucose on the Fc N-glycan. Afucosylated antibodies bind FcγRIIIa with substantially higher affinity than fucosylated counterparts, enhancing ADCC activity by 5- to 50-fold. For antibodies where ADCC is the mechanism of action, afucosylation is a key CQA that must be characterized and controlled within defined acceptance limits.
Does every therapeutic antibody need glycan analysis in its release specification?
Approximately 50% of FDA-approved biologics include glycan analysis in drug substance release specifications. For antibodies with ADCC or CDC activity, glycan analysis is standard and often combined with functional bioassays. For antibody fragments produced in E. coli or antibodies engineered to be aglycosylated, glycan analysis may not be required in release specifications, but the decision must be justified.
How is glycan comparability assessed for biosimilars?
Biosimilar developers must characterize the glycan profile of both the proposed biosimilar and the reference product across multiple lots. Statistical acceptance criteria are pre-defined based on reference product variability. Functional bioassays (ADCC, CDC, FcγR binding) are used to confirm that any observed glycan differences do not translate into meaningful changes in biological activity.
What is the totality-of-evidence approach to glycan assessment?
The totality-of-evidence approach means integrating structural glycan characterization data with functional bioassay results, PK data, and clinical context to support a comparability or biosimilarity conclusion. No single assay result is interpreted in isolation. The FDA has consistently applied this framework in biosimilar reviews and process change comparability assessments.
What analytical methods are recommended for glycan profiling?
Released N-glycan profiling using HILIC with fluorescent labeling (2-AB or RapiFluor-MS) is the most common approach in BLA submissions. LC-MS glycopeptide analysis and intact mass analysis provide orthogonal structural confirmation. Lectin-based methods are emerging as complementary tools for rapid glycan epitope profiling of intact antibodies.
How does the expression system affect the glycan profile?
CHO cells produce primarily complex-type, core-fucosylated glycans with very low sialylation. NS0 and SP2/0 cell lines produce non-human glycans (alpha-Gal and Neu5Gc, respectively) that can be immunogenic. Fc fusion proteins often exhibit higher sialylation and more complex multi-antennary structures compared to standard mAbs. Expression system selection directly shapes the glycan profile and must be addressed in characterization.
References
Jefferis, R. (2009). Glycosylation as a strategy to improve antibody-based therapeutics. Nature Reviews Drug Discovery, 8, 226–234. https://doi.org/10.1038/nrd2804
Sola, R.J., & Griebenow, K. (2010). Glycosylation of therapeutic proteins: an effective strategy to optimize efficacy. BioDrugs, 24(1), 9–21. https://doi.org/10.2165/11530550-000000000-00000
ICH Q8(R2). (2009). Pharmaceutical Development. International Council for Harmonisation. https://www.ich.org/page/quality-guidelines
Shields, R.L., et al. (2002). Lack of fucose on human IgG1 N-linked oligosaccharide improves binding to human FcγRIII and antibody-dependent cellular toxicity. Journal of Biological Chemistry, 277(30), 26733–26740. https://doi.org/10.1074/jbc.M202069200
Zhang, B. (2026). Antibody Glycosylation Insights for High-Quality Biotherapeutics. FDA Grand Rounds, January 22, 2026. Office of Pharmaceutical Quality (OPQ), Center for Drug Evaluation and Research (CDER), FDA. https://www.youtube.com/watch?v=INjCbBVkuVE
Goetze, A.M., et al. (2011). High-mannose glycans on the Fc region of therapeutic IgG antibodies increase serum clearance in humans. Glycobiology, 21(7), 949–959. https://doi.org/10.1093/glycob/cwr027
Chung, C.H., et al. (2008). Cetuximab-induced anaphylaxis and IgE specific for galactose-alpha-1,3-galactose. New England Journal of Medicine, 358(11), 1109–1117. https://doi.org/10.1056/NEJMoa074943
Konno, Y., et al. (2012). Influence of cell culture conditions on IgG glycosylation. Journal of Bioscience and Bioengineering, 113(4), 481–487. https://doi.org/10.1016/j.jbiosc.2011.11.021
Luo, S., & Zhang, B. (2024). Benchmark glycan profile of therapeutic monoclonal antibodies produced by mammalian cell expression systems. AAPS Journal (in press). See also: FDA. (2024). Novel method for rapid glycan profiling of therapeutic monoclonal antibodies. https://www.fda.gov/drugs/regulatory-science-action/novel-method-rapid-glycan-profiling-therapeutic-monoclonal-antibodies
ICH Q6B. (1999). Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products. International Council for Harmonisation. https://www.ich.org/page/quality-guidelines
ICH Q5E. (2004). Comparability of Biotechnological/Biological Products Subject to Changes in Their Manufacturing Process. International Council for Harmonisation. https://www.ich.org/page/quality-guidelines
European Medicines Agency. (2012). Guideline on development, production, characterisation and specifications for monoclonal antibodies and related products. EMA/CHMP/BWP/532517/2008. https://www.ema.europa.eu
Duivelshof, B.L., et al. (2019). Glycan-based strategy for biosimilar characterization. Analytical and Bioanalytical Chemistry, 411(27), 7291–7305. https://doi.org/10.1007/s00216-019-01937-1



