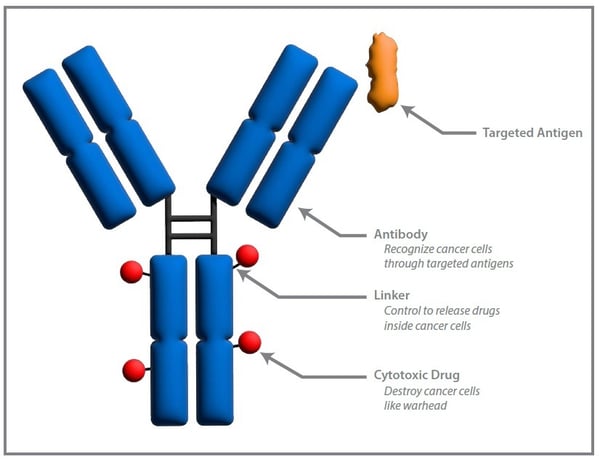
Biologics Characterization and CMC Analytical Services
Our CMC analytical services cover the full characterization lifecycle, from early development through clinical stages. We support monoclonal antibodies, antibody drug conjugates, recombinant proteins, bispecific antibodies, biosimilars, and nucleic acids across all development stages.

When programs need an expert on CMC analytics
Development teams typically engage us when:
- Conducting a CMC gap analysis to identify missing or insufficient analytical data before regulators do
- Preparing CMC analytical packages for IND submission and regulatory interactions
- Transitioning from early development into clinical stage manufacturing
- Managing manufacturing changes, comparability exercises, or biosimilar similarity strategies
- Investigating complex analytics, such as stability or heterogeneity
Our role is to determine whether the current analytical framework can withstand regulatory scrutiny. If not, we reinforce it by clarifying critical quality attributes, closing analytical gaps, strengthening control strategies, and structuring comparability data, so it remains scientifically coherent and defensible during review.
How regulators evaluate biologics characterization and CMC analytical
During IND and BLA review, agencies assess whether analytical data clearly define and control critical quality attributes (CQAs).
Review focuses on whether attributes are:
- Scientifically sound and reliably measurable using qualified or validated analytical methods
- Controlled within clearly defined and defensible limits
- Mechanistically interpretable across the product lifecycle, including after manufacturing changes
Analytical gaps often trigger information requests when impurity limits lack justification, stability trends are unclear, or structural heterogeneity is insufficiently characterized.
To avoid that, CMC analytical strategies should be defined early, supported by orthogonal methods, and aligned with a clear control framework that links structure, function, and manufacturing consistency.

Crystal Bio Solutions CMC Analytical Capabilities
We operate a 5,300 sq ft CMC analytical laboratory in New Jersey, providing complex biologics characterization services across three analytical pillars. Key platforms and methodologies include:
| Chromatographic Techniques | Electrophoretic & Biophysical Methods | High-Resolution Mass Spectrometry |
| Size-exclusion chromatography (SEC) for aggregation analysis | Capillary Electrophoresis SDS (CE-SDS) for purity and fragmentation | Intact mass & peptide mapping for sequence and PTM analysis, including charge variants, disulfide, and glycosylation characterization |
| Ion exchange chromatography (IEX) for charge variant profiling | Dynamic light scattering (DLS) for particle sizing | Serum stability, in vivo CQA analysis, and biotransformation studies |
| Hydrophobic interaction chromatography (HIC) for ADC characterization | Differential scanning calorimetry (DSC) for thermal stability | Biosimilar comparability |
| Reverse-phase chromatography (RP) for purity assessment | UV and fluorescence spectroscopy for structural monitoring |
Diverse antibody formats and conjugates, including multispecifics, ADCs / AOCs, and other complex modalities |
| FTIR for secondary structure assessment | CMC gap analysis |
How we characterize antibody-based drugs
Monoclonal antibodies and antibody-derived formats require structured analytical control due to their inherent heterogeneity. CMC analytical development defines the molecule through sequence confirmation, structural variant mapping, impurity control, and stability assessment, establishing the critical quality framework reviewed during IND and BLA evaluation.
For programs where in vivo behavior requires deeper investigation, we perform full protein characterization directly in biological matrix using specialized sample preparation coupled with LC-MS. This enables serum stability analysis and troubleshooting of PK samples from animal and human studies.
Teams often struggle when CQAs are not defined early, whether because development timelines compress analytical work or because characterization is deferred until after manufacturing scale-up. In antibody-drug conjugates, DAR distribution, conjugation variability, and payload stability add further complexity. The result can be unstable comparability after process changes, unexpected immunogenicity signals, or regulatory information requests during review..
Biosimilar drug development in 2026
Biosimilar drugs are biologic medicines developed to be highly similar to an approved reference product, with no clinically meaningful differences in safety, purity, or potency. Recent FDA approvals show a clear direction: analytical similarity now drives biosimilar development, with reduced reliance on large comparative efficacy trials when structural and pharmacokinetics (PK) evidence is strong.
However, projects often present incomplete glycan characterization, weak functional assay design, and poorly justified similarity ranges, which can trigger FDA information requests, delay PK studies, force repeat analytical work, narrow similarity margins, or ultimately require additional clinical confirmation, increasing cost and extending approval timelines.
Frequently Asked CMC Questions
What is CMC analytical development for biologics?
CMC analytical development covers method design, qualification, and validation across identity, purity, potency, and stability. It produces the data packages that support IND and BLA submissions and define the control strategy for the product lifecycle.
What differentiates method development from analytical strategy?
Method development generates data. Analytical strategy defines which attributes matter, how they are justified, and how they support regulatory conclusions.
What is a CMC gap analysis and when should it be conducted?
A CMC gap analysis reviews the current analytical package against regulatory expectations to identify missing methods, insufficient validation, or poorly justified limits. It should be conducted at least six to nine months before IND submission, while there is still time to close gaps without delaying the filing.
Which critical quality attributes are most commonly underdefined in early biologic programs?
Glycosylation, charge variants, and aggregation are frequently undercharacterized in early development because they are deprioritized during lead selection. These attributes directly affect immunogenicity risk, pharmacokinetics, and comparability interpretation, making late-stage gaps difficult and costly to resolve.
How does CMC analytical development influence IND review timelines?
Incomplete structural definition, poorly justified impurity limits, or unclear stability interpretation frequently generate regulatory information requests. Early alignment of analytical data with regulatory expectations reduces submission risk and review delays.
When does a manufacturing change require expanded comparability analysis?
Changes in scale, cell line, purification steps, raw materials, or formulation can affect critical quality attributes. Expanded orthogonal characterization is required when attribute variability increases or process risk shifts.
What level of analytical similarity is expected for biosimilars?
Regulators expect extensive structural and functional similarity supported by orthogonal methods and statistical evaluation. Analytical data must justify reduced clinical burden within the totality-of-evidence framework.
How does CBS support complex biologics characterization for ADCs and other advanced modalities?
CBS applies orthogonal analytical platforms including LC-MS, HIC, and cell-based assays to characterize ADC-specific attributes such as DAR distribution, conjugation site occupancy, and payload stability. The same infrastructure supports mRNA integrity, LNP encapsulation efficiency, and AAV capsid characterization for advanced modality programs.